New Study Points to Degradable Commodity Plastics Without Changing Manufacturing
Apr 9, 2026
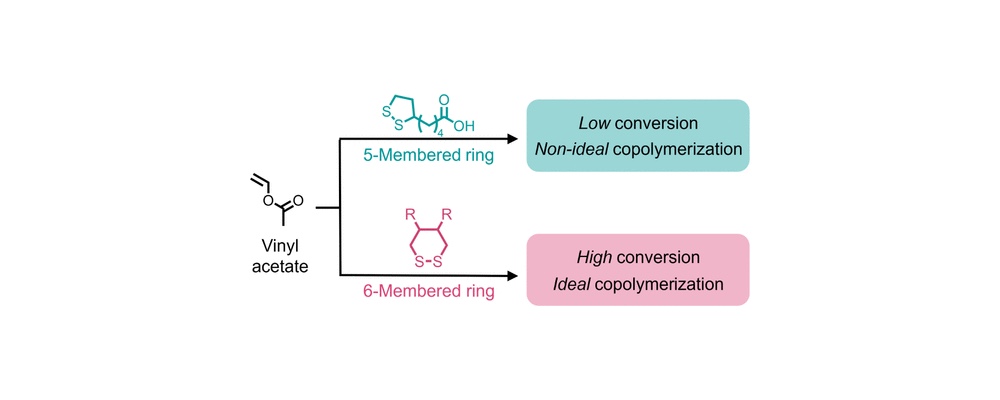
Researchers from UC Santa Barbara’s BioPACIFIC MIP have reported a new route to making commodity plastics more sustainable without requiring major changes to existing manufacturing methods. In a recent paper published in the Journal of the American Chemical Society, the team showed that six-membered cyclic disulfides known as 1,2-dithianes can be efficiently copolymerized with less-activated monomers such as vinyl acetate under simple thermal free-radical conditions.
The study found that the approach delivered high conversions of both monomers above 90 percent, tunable molar masses, and scalability beyond 20 grams. The resulting copolymers also incorporated cleavable sulfur-containing units into the polymer backbone, creating a practical path toward degradable vinyl materials derived from inexpensive, industrially relevant feedstocks.
Beyond processability, the materials showed major performance gains. The paper reports that incorporating the new cyclic disulfide chemistry into vinyl acetate systems can raise toughness by roughly 4000 percent relative to poly(vinyl acetate) alone, while allowing properties to span from semicrystalline plastics to elastomeric materials.
The work, led by Parker T. Morris and co-authored by Sarah C. Olsen, Marvin Santiago, Hanlin M. Wang, Elliott H. Denton, Javier Read de Alaniz, Christopher M. Bates, Kaitlin R. Albanese, and Craig J. Hawker, highlights how small changes in monomer design could help bring degradable versions of widely used plastics closer to commercial reality.